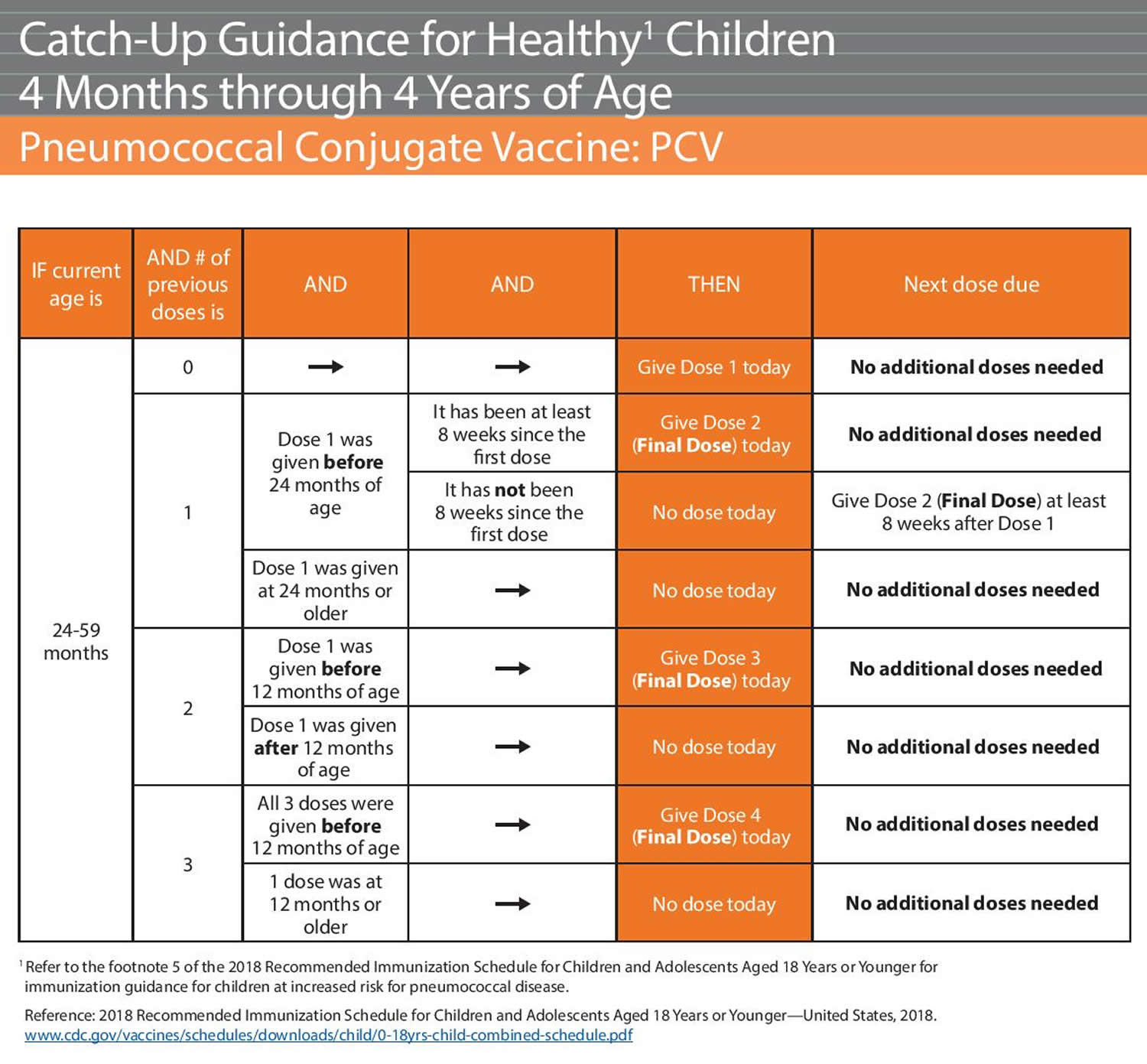
The disparate results reflect different inclusion and exclusion criteria for studies and designs. 
However, the estimated collected body of evidence on pneumococcal pneumonia varied greatly between the reviews-some reviews indicated a statistically significant VE, while others did not ( Table S1). The pooled VE on invasive pneumococcal disease was estimated as 73% in randomized controlled trials (RCTs) and as 45–59% in observational studies ( Table S1). 
In 20, five different systematic reviews evaluated the vaccine efficacy and effectiveness (hereafter both will be abbreviated VE) of the PPV23 vaccine ( Table S1). The study was conducted in the Netherlands, included almost 85,000 participants, and found a modified intention-to-treat vaccine efficacy of 37.7% (95% CI: 14.3 to 55.1) against the first episode of vaccine-type community acquired pneumococcal pneumonia (VT-CAP) and 75.8% (95% CI: 46.5 to 90.3) against the first episode of vaccine-type invasive pneumococcal disease (VT-IPD). The randomized placebo-controlled trial named CAPITA demonstrated the efficacy of PCV13 in adults aged ≥ 65 years. With the exception of serotype 6A, all serotypes in PCV13 are also included in PPV23. In an effort to reduce this burden, two different vaccines have been developed - a 23-valent polysaccharide vaccine (PPV23, Pneumovax 23, Sanofi Pasteur, MSD) and a 13-valent conjugate vaccine (PCV13, Prevenar13, Pfizer). Estimates show that the number of hospitalizations due to pneumococcal pneumonia will double in the US from 2004 to 2040 without any interventions, and that this will lead to a $ 2.5 billion increase in health care expenditure. pneumoniae can cause non-invasive disease such as pneumonia, sinusitis and otitis media. The most severe form of pneumococcal disease is invasive pneumococcal disease (IPD). Streptococcus pneumoniae is a leading cause of mortality and morbidity in the elderly population. 
Some of the results indicate that the effectiveness of the PPV23 is best in younger age groups, and that it decreases over time. Our estimates for the protective vaccine efficacy and effectiveness (VE) of PPV23 on pneumonia and pneumococcal pneumonia overlap with results from previously published reviews. New high-quality observational studies indicate protective vaccine effectiveness for both vaccines against vaccine type pneumonia. Due to different outcomes, it was not possible to do a meta-analysis. No new randomized clinical trials were identified. We identified nine studies on PCV13 and six on PPV23. Results were stratified by vaccine type and outcome. Risk of bias was assessed using Cochrane Risk of Bias tool for and the Newcastle–Ottawa Scale. We systematically searched for clinical trials and observational studies published between Januand Apin Pubmed, Embase, Cinahl, Web of Science, Epistemonikos and Cochrane databases. The objective of this review is to update the evidence base for vaccine efficacy and effectiveness of PPV23 and PCV13 against invasive pneumococcal disease and pneumonia among an unselected elderly population. Two different vaccines are available for the prevention of pneumococcal disease in adults: a 23-valent polysaccharide vaccine (PPV23), and a 13-valent conjugated vaccine (PCV13). Adult vaccination is high on the agenda in many countries.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
